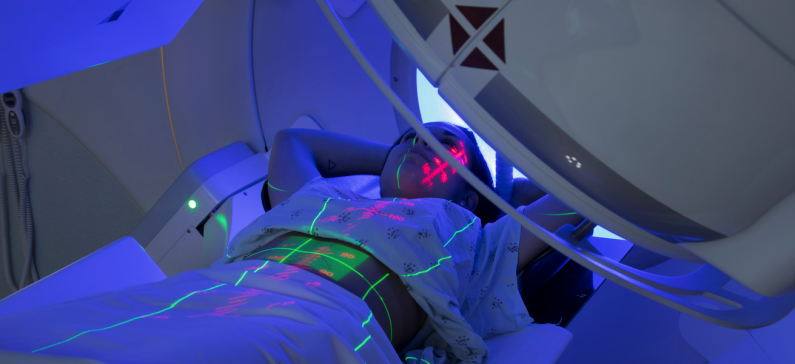
New “one second” method of radiation therapy for cancer patients
A shorter and less expensive way to treat cancer patients with radiation opens the pioneering research of scientists in the US, headed by oncologist of Greek origin. In the future, cancer patients will probably be able to receive all their radiation therapy, which now lasts several weeks, in a single dose of only a few milliseconds.
The new express-radiation FLASH has begun to develop at the University of Pennsylvania Medical School’s Abramson Cancer Center, and the researchers, led by oncologist Costas Koumenis, have conducted animal experiments on related animals. International Journal of Research Oncology, Biology & Physics.
Dr. Koumenis is a graduate of the Aristotle University of Thessaloniki (1989), with a doctorate from the University of Houston in Texas (1994), while Ioannis Verginadis and Anastasia Velalopoulou participate in the research team. Scientists have used proton radiation to create an accelerator at a dose of 100 to 200 milliseconds that -theoretically at least- is enough to give a cancer patient all the necessary radiation. Early trials of the FLASH technique show that it has the same effect on tumors as traditional photon therapy, without actually damaging healthy tissues because of its very short duration.
Other scientific groups have created similar doses of express, but using low-energy electrons, which do not penetrate deep enough into the body (only five to six inches) to have the same therapeutic utility for internal tumors. The new technique (Proton FLASH Radiation Therapy System), tested at the Roberts University Proton Therapy Center, shows that, with appropriate technical modifications, current proton accelerators can achieve doses of clinical utility.
The researchers are already preparing for their first human clinical trial. So far, the FLASH technique has shown its efficacy only in small animals and small tissue areas. The method needs to be refined to prove to have a similar effect on a larger area of the body and on larger bodies such as the human. In addition, the dose will be sought to last less than 100 milliseconds.
It is noteworthy that scientists have not yet understood exactly how express-radiation proton-bombardment works and, most importantly, how it manages to do no damage to adjacent healthy tissues. It is not clear why cancer cells respond differently to healthy ones, resulting in the former being destroyed but the latter intact.
Scientists estimate that proton-expressing radiation or high-energy electrons will be clinically available in five to ten years, even for tumors that are currently difficult to treat with radiation. But maybe after two to three years it can only be used for superficial tumors and for those revealed after surgery.









Innovation
-18/03/2022 2:51 am
In particular, the origin of radiotherapy dates back to the late XIX century, with the discovery of X-rays by Wilhelm Conrad R ntgen (R ntgen, 1896 ; Busacchi, 2015 ). In the following years, Marie and Pierre Curie identified a substance with radiations two million times higher than Uranium (studied by Becquerel), that they called Radium (1898) (Ku akowski, 2011 ). The two scientists initially studied the use of X-rays for diagnostic purposes but eventually realized that they were harmful at the cellular level, thus suggesting their use in the treatment of tumors (Curie and Curie, 1899 ). X-rays were already used in 1896 by Emil H. Grubb for the treatment of breast cancer (Grubb , 1933 ; Nakayama and Bonasso, 2016 ), while Anton Ultimus Sj gren applied this treatment to an epithelioma of the mouth in 1899 (Nakayama and Bonasso, 2016 ). However, modern radiotherapy only began in 1920, when Claudius Regaud demonstrated that radiation fractionation could be used to treat several human cancers, by reducing the side effects of the treatment itself (Deloch et al., 2016 ; Moulder and Seymour, 2017 ). Despite these achievements, medical and interventional approaches to tumors before the Second World War were essentially radical methods, aimed at the complete eradication of the disease before it can spread and metastasize throughout the organism. Therefore, surgical treatment, often representing the only therapeutic option despite its demolition impact, resulted ineffective in patients with advanced tumor pathology or whenever the surgical act failed to remove all the tumor mass (Hajdu, 2011 ; Hajdu and Vadmal, 2013 ). An epochal turning point for the treatment of tumors was reached in the mid-19, with the birth of chemotherapy and the subsequent evolution of modern medical therapy of tumors. The discovery and synthesis of new compounds represented the basis to develop effective therapeutic interventions in patients with different types of advanced solid tumors or hematological drug treatments, alone or in association with surgical and radio treatments (Arruebo et al., 2011 ; Figure Figure2